Defective Products and Dangerous Drugs
Although the manufacturing and marketing of prescription drugs and other products are regulated, there are still some on the market that cause more harm than good, and sadly, the injuries caused by these drugs and products can be permanent, if not life-threatening. Suffering an injury as a result of faulty drugs and products should never happen–but it does.
At Richard Harris Personal Injury Law Firm, our Las Vegas defective product lawyers work hard to make sure our clients are treated fairly when they’ve been injured as a result of someone else’s negligence.

Camp Lejeune Drinking Water Contamination
From 1953 to 1987 the Marines and their families, along with civilian employees at Camp Lejeune may have been exposed to contaminated drinking water.
Talcum Powder
Talcum powder is seemingly as innocuous a product as can be imagined. The product has been in households in one form or another, since before most of us were born. We may have been exposed to it as infants in baby powder, it’s most common form.
Allergan BIOCELL® Textured Breast Implant and Tissue Expander Recall
In the past two decades, some women have developed Anaplastic Large Cell Lymphoma (ALCL), a rare form of cancer, from breast implants.

Roundup Weed Killer Linked to Cancer
Roundup is the most widely used herbicide in homes and farms throughout the world. However, exposure to the weed killer’s primary ingredient, glyphosate, has been linked to deadly forms of cancer, including non-Hodgkin lymphoma.

E-Cigarette – JUUL Products
JUUL Labs, the maker of the JUUL e-cigarette device, accounts for approximately 75% of e-cigarettes sold in the United States.

Walk-in Bathtubs for the Elderly
A variety of whirlpool/Jacuzzi-style tub manufacturers aggressively market their expensive products to the elderly, citing increased safety and other health benefits. Unfortunately for older Americans and their adult children, scientific research fails to back up these claims. Instead, statistics show that more and more elderly people are suffering severe or fatal injuries after falling in poorly designed tubs they cannot escape from.

Military Earplug Hearing Loss
The government issues earplugs to help protect military service members from hearing loss during combat and training. One particular model—known as the 3M Combat Arms earplug—is shown to be defective, which places service members at unnecessary risk of permanent hearing loss and/or tinnitus.
Onglyza
Onglyza aids in the maintenance of type 2 diabetes. Patients often use Onglyza as a part of a complete treatment program including diet, exercise and weight management. Onglyza helps control high blood sugar and regulates the levels of insulin the body produces.
Invokana
During this first year period, the Institute for Safe Medication Practices (ISMP), a pharmaceutical industry watchdog organization, and publisher of the pharmaceutical industry review Quarterwatch, found 457 “serious adverse events” reported.
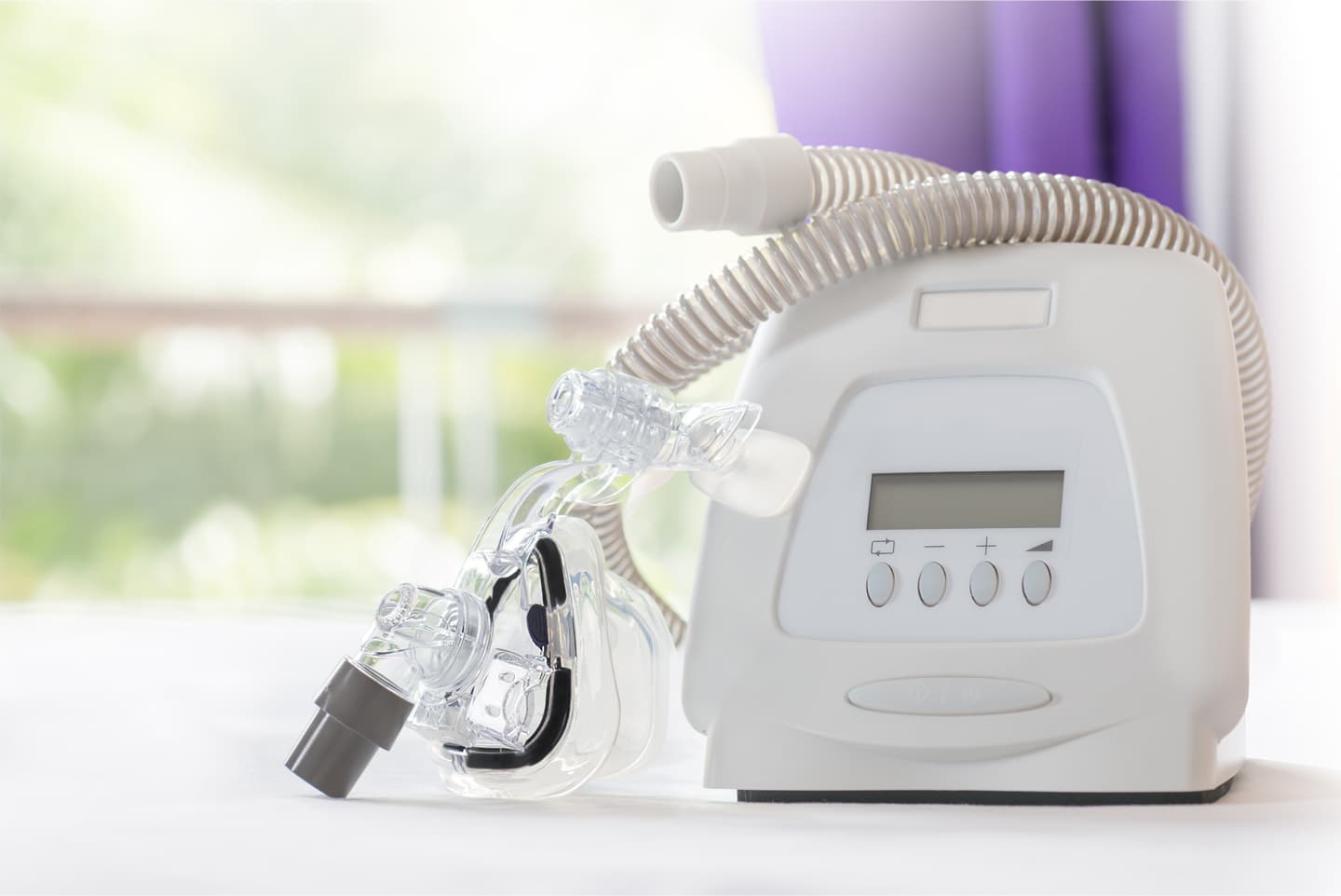
PHILIPS CPAP and BiPAP Ventilator Devices
Philips sleep and respiratory care devices have been recalled after it was discovered that the sound abatement foam that is used in these products is linked to respiratory illness and lung injury.


















